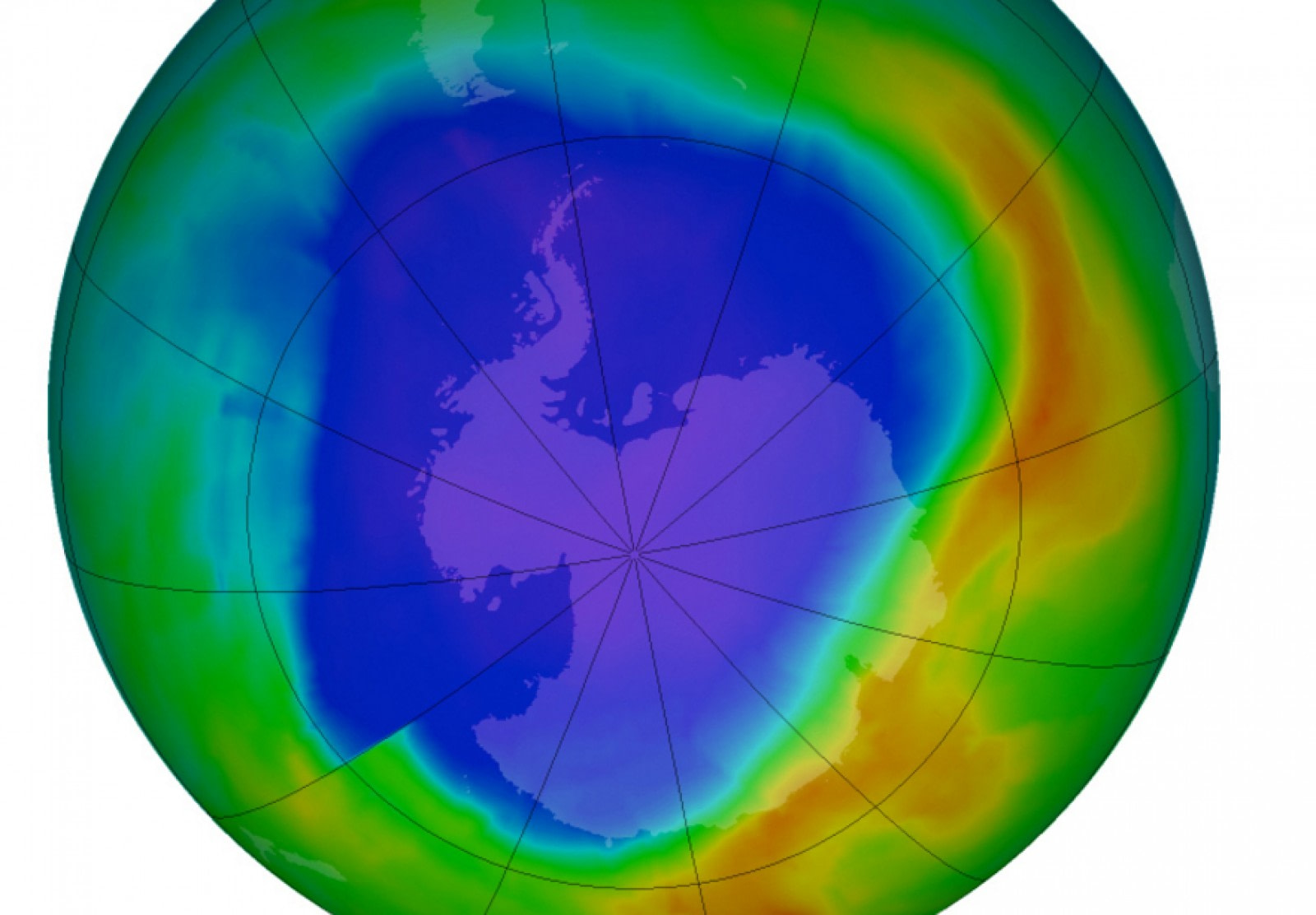
The ozone layer in Antarctica
An ozone molecule consists of three oxygen atoms instead of the usual two. It exists only in the atmosphere in trace quantities. Ozone molecules are formed by the interaction of ultraviolet radiation (UV) from the sun with oxygen molecules: When an O2 molecule is split the two free oxygen atoms bond with other o2 molecules to form O3 molecules.
Because UV radiation is intense at higher altitudes where the air is thinner it is in the stratosphere where most of the ozone is produced: Scientists have calculated that if the ozone layer was brought down to sea level it would be 3 mm thick, while in the Antarctic the ozone can be as low as 1 mm thick. Ozone in the stratosphere is being depleted by a variety of human-made gases with a ‘hole’ forming over Antarctica.
The ozone hole over Antarctica
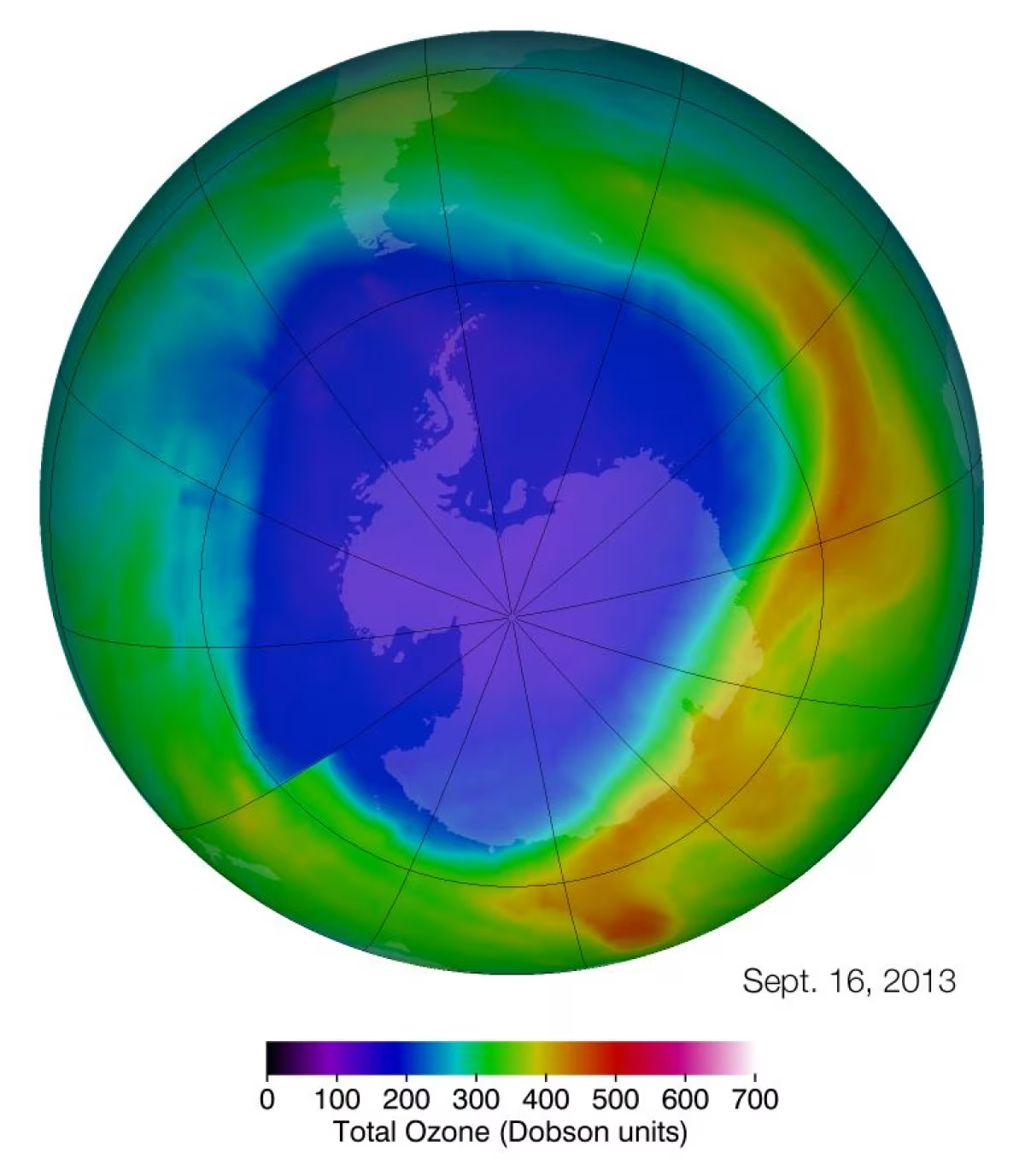
By NASA's Goddard Space Flight Center [Public domain], via Wikimedia Commons
The ozone hole is one of the largest impacts humans have had on Antarctica. From the 1940s to the 1990s rapid industrialisation and higher living standards have led to chlorofluorocarbons, hydrochlorofluorocarbons, halons and methyl bromide creating a ‘hole’ in the ozone over the Antarctic continent.
The ozone hole was detected by scientists when they compared the amount of ozone found in the early 1980s with measurements dating back to 1956. The hole was found to vary in size with the hole forming during the two months of September and October. Each winter a polar vortex forms in the stratosphere over Antarctica with temperatures plummeting to as low as – 85° Celsius in the lower atmosphere. At these low temperatures ice clouds form and act as sites where chlorine and bromine-containing chemicals are converted to compounds that destroy ozone.
Study of clouds key to understanding the ozone hole
Specifically, polar stratospheric clouds (PSCs) provide surfaces for chemical reactions to take place involving chlorine which destroys ozone molecules. PSCs form around 60°S latitude in the altitude range of 10-25 kilometres during the winter and early spring. The clouds are classified into Type I and Type II depending on their particle size and formation temperature.
Type I PSCs are much thinner than Type II clouds and have a formation threshold temperature of 5 to 8° Celsius above freezing. These clouds consist mainly of droplets of nitric acid and sulphuric acid while Type II clouds, also known as nacreous or mother-of-pearl clouds, are composed of ice crystals and form when temperatures are below the ice-frost point (usually below – 83°C.
Despite decades of research there is still gaps in knowledge on PSCs for example the timing and duration of PSC events, their geographic extent and vertical distributions, which impacts the accuracy of ozone depletion models. In the spring time, when sunlight returns to Antarctica, the destruction of the ozone within the polar vortex begins, reaching a maximum in early October then declines over the period to end of December.
The ozone layer protecting us from harmful radiation
The majority of ozone in the Earth’s atmosphere lies between 15 and 30 km in altitude: in the stratosphere where it absorbs harmful radiation from the sun:
Around 2 percent of the light the sun emits is in the form of high-energy ultraviolet (UV) radiation with some of this UV radiation (UV-B) causing damage to living things including sunburn, skin cancer and eye damage. The amount of sola UV radiation that reaches Earth at any location is dependent on the position of the sun above the horizon, the amount of ozone in the atmosphere and local cloudiness and pollution levels. With an ozone hole forming over Antarctica scientists large increases in UV-B during the Antarctic spring.
Global agreement to halt ozone-destroying chemicals
Since the Montreal Protocol was signed in 1989, in which deadlines for reducing and eliminating the production and use of ozone depleting substances, there has been a significant reduction in ozone destroying chemicals in the atmosphere with the ozone layer over Antarctica expected to return to 1980 levels sometime in the latter half of the 21st century. Nonetheless, the timing of the recovery is uncertain due to uncertainty in models used to project future changes.
The ozone hole getting smaller in size
In a recent study the ozone hole found over your head during an Antarctica expedition was found to be showing evidence of a decrease in size based on 15 years of ground and satellite observations with the size of the ozone hole in 2014 less severe compared to the 1995-2005 average. The study found the ozone hole to be 20.9 square kilometres, the sixth smallest over the 1991-2004 period with the data showing that since 1998 the ozone hole is getting smaller at a rate of 0.17 square kilometres per annum.
However, what is causing the ozone hole to shrink is not clear. Antarctic stratospheric ozone depleting substances are estimated using equivalent effective stratospheric chlorine (EESC), which is a combination of chlorine and bromine. A mean average of 5.2 years is used to calculate EESC and since the 2000-2002 peak of 3.70 parts per billion (ppb) EESC has decreased to 3.49 ppb: a decrease of 0.34 ppb or 9%. This means that EESC levels have dropped by 20% towards the 1980 level of 2.05 ppb where 1980 is considered by researches as the ‘pre-ozone hole period’.
The link between the ozone layer and climate
The Australian Antarctic Division has been conducting research on how the recovery of the ozone layer will produce significant feedbacks to the surface climate of Antarctica and the Southern Hemisphere during the remainder of the 21st century, in terms of changes in seasonal cycles and long term temperature and wind trends.
To gain greater understanding the Australian Antarctic Division is developing a chemistry climate model through the Australian Community Climate Earth System Simulator (ACCESS). ACCESS incorporates the United Kingdom Chemistry and Aerosols model (UKCA). The prime objective of Australia’s ACCESS project is to provide analyses and advice on the effects of feedbacks from ozone recovery on Southern Hemisphere climate. Specifically the project seeks to fully incorporate the UKCA chemical model into the ACCESS model and compare outputs to standard scenarios with historical observations on a regional scale.
Australian research on the ozone layer
The expected outcomes of the ACCESS programme is the implementation of a new Earth system modelling capability for Australia and peer-reviewed analysis on changes in Antarctic and Southern Hemisphere climate processes for scientific journals and advice to government agencies.
In addition, the project is expected to foster increased trans-Tasman cooperation between Australia and New Zealand: New Zealand’s NIWA has been conducting research on Antarctica’s ozone hole as it has a major effect on local climate, which in turn influences global climate and sea level changes.
In particular NIWA’s modelling of the atmospheric chemistry of feedback processes between stratospheric ozone and the Antarctic climate will increase the accuracy of global climate change models. Up to then Australia’s model focused on weather predictions and climate projects and did not incorporate interactive stratospheric chemistry.
In addition, ACCESS aims to increase cooperation between Australian and international scientists on chemistry-climate modelling and the regional effects of ozone recovery on the Southern Hemisphere. In addition, the Australian Antarctic Division has been conducting research on PSCs with the Davis Station LIDAR being used since 2001 to study stratospheric clouds. The measurements are being used to investigate the climatology of the clouds and their relation to the temperature structure of the stratosphere.